Introduction of Symbiotic and Nonsymbiotic Nitrogen Fixation
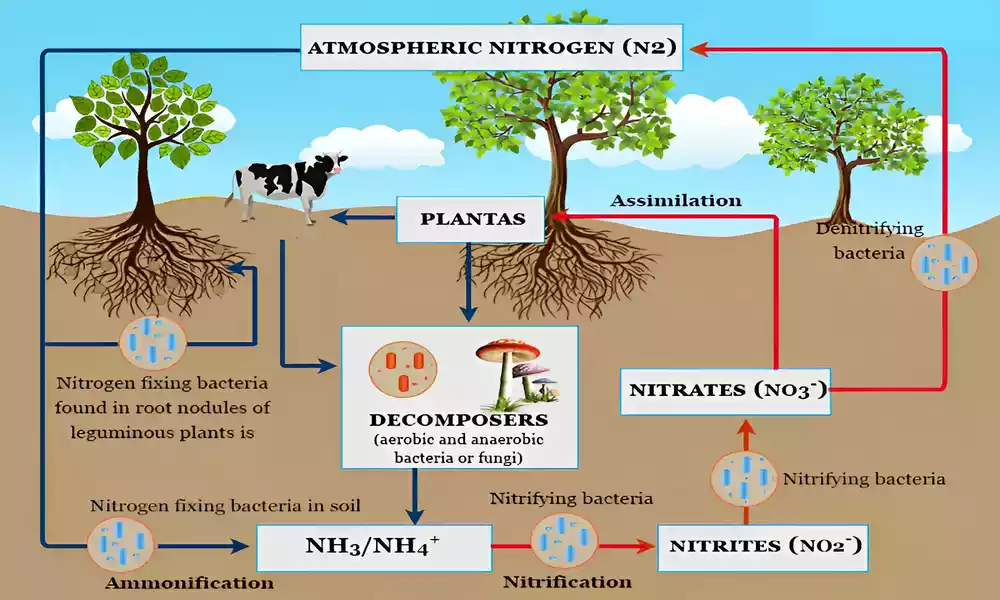
Nitrogen is essential for all living organisms, playing a crucial role in the structure of proteins, nucleic acids, and other biomolecules. Despite its abundance in the Earth’s atmosphere, atmospheric nitrogen (N2) is largely inert and cannot be directly utilized by most organisms.
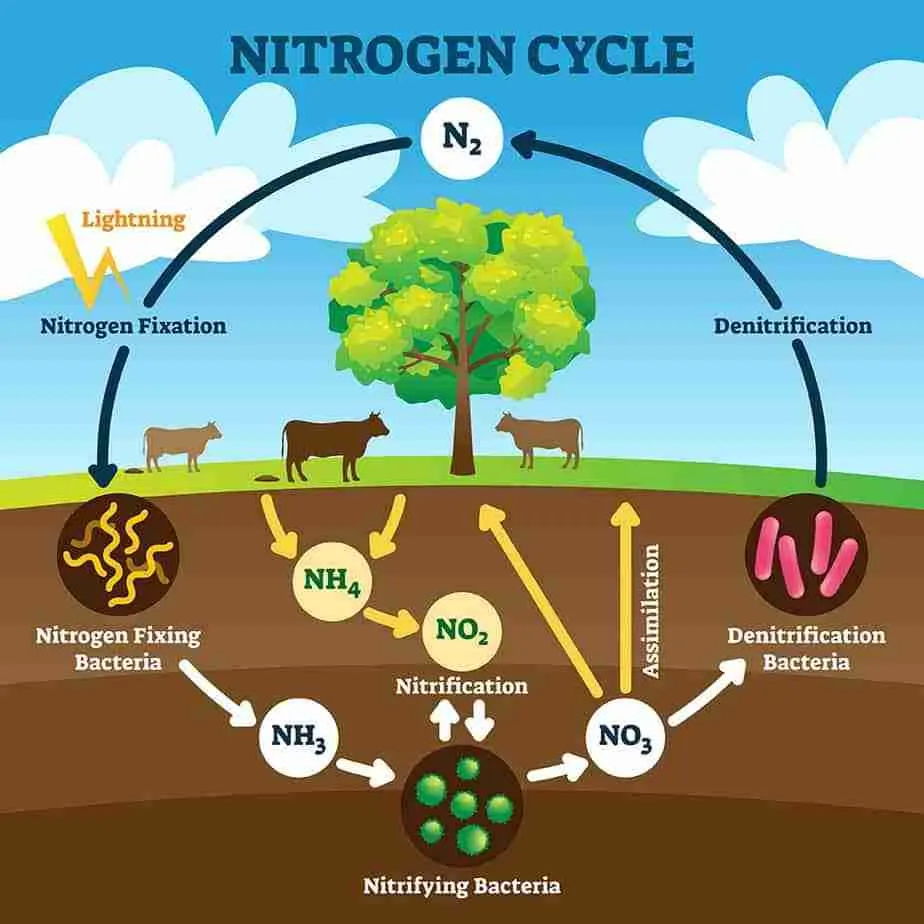
Nitrogen fixation is the process by which atmospheric nitrogen is converted into forms that are biologically accessible and usable. This conversion is carried out by nitrogen-fixing microorganisms, which play a pivotal role in making nitrogen available to plants and, consequently, to higher trophic levels in the food chain.
Symbiotic Nitrogen Fixation
Symbiotic nitrogen fixation is a biological process in which certain plants form specialized partnerships with nitrogen-fixing microorganisms to convert atmospheric nitrogen (N2) into biologically usable forms, such as ammonia (NH3) or nitrate (NO3-).
This process is facilitated by a close interaction between the plant and the nitrogen-fixing microorganism, typically bacteria, which benefits both partners and has significant ecological and agricultural implications. Two prominent examples of symbiotic nitrogen fixation include the legume-rhizobium association and actinorhizal symbiosis.
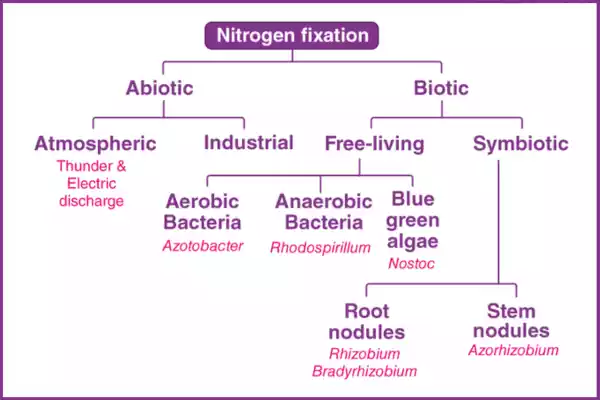
- Legume-Rhizobium Association:
- Process of Nodulation: Leguminous plants, like beans, peas, and clover, form nodules on their roots as a result of a mutualistic relationship with nitrogen-fixing bacteria known as rhizobia. These bacteria infect the root cells of the plant, forming nodules where they reside.
- Nitrogen Fixation: Inside the nodules, rhizobia differentiates into bacteroids, which are specialized nitrogen-fixing forms. The plant supplies energy in the form of carbohydrates to the bacteroids, and in return, the bacteroids convert atmospheric nitrogen into ammonia through the process of nitrogen fixation.
- Role of Leghemoglobin: To create a low-oxygen environment favorable for nitrogen fixation, leguminous plants produce a protein called leghemoglobin within their nodules. Leghemoglobin binds oxygen, preventing it from inhibiting the nitrogen-fixing enzymes.
- Regulation of Nitrogen Fixation Genes: Both the plant and the rhizobia regulate the expression of genes involved in nitrogen fixation, ensuring that resources are allocated efficiently.
- Actinorhizal Symbiosis:
- Examples of Actinorhizal Plants: Certain woody plants, such as alder and bayberry, engage in actinorhizal symbiosis. These plants associate with a group of nitrogen-fixing bacteria called Frankia.
- Role of Frankia Bacteria: Frankia bacteria penetrate root cells, forming structures similar to nodules called “actinorhizal nodules.” Inside these nodules, the bacteria fix atmospheric nitrogen.
Symbiotic Nitrogen Fixation Benefits and Drawbacks:
Advantages for Plants:
- Access to Usable Nitrogen: Plants receive a direct nitrogen source in a form they can readily use for growth and development.
- Reduced Nitrogen Demand: Symbiotic nitrogen fixation reduces the plant’s reliance on external nitrogen sources like soil nitrates or fertilizers.
- Energetic Efficiency: The energy invested by the plant in providing carbohydrates is often more efficient in terms of nitrogen gain than absorbing nitrates from the soil.
Environmental Impacts:
- Enhanced Soil Fertility: Nitrogen fixation enriches the soil with nitrogen compounds, benefitting other non-nitrogen-fixing plants in the vicinity.
- Reduced Fertilizer Dependency: Symbiotic nitrogen fixation can reduce the need for nitrogen-based fertilizers, mitigating nutrient pollution.
Challenges and Limitations:
- Energy Cost: The plant invests energy in supporting the microorganisms, which might affect its overall growth and reproduction.
- Nutrient Competition: The plant and microorganisms must balance their nutrient requirements, as competition for resources can impact the relationship.
- Specificity: Symbiotic relationships are often species-specific, limiting the range of plants that can benefit from a particular microorganism.
Symbiotic nitrogen fixation is a prime example of the intricate ecological relationships that shape ecosystems. It highlights the interconnectedness of organisms and showcases how biological partnerships can drive nutrient cycling, soil fertility, and overall ecosystem health.
Nonsymbiotic Nitrogen Fixation
Nonsymbiotic nitrogen fixation is a process by which certain microorganisms, primarily bacteria, convert atmospheric nitrogen (N2) into biologically usable forms, such as ammonia (NH3) or nitrate (NO3-), without forming specialized partnerships with plants.
Unlike symbiotic nitrogen fixation, which involves close interactions between specific plant hosts and nitrogen-fixing microorganisms, nonsymbiotic nitrogen fixation occurs independently in the soil, water, or even within certain organisms. This process has both natural and human-induced mechanisms and plays a significant role in global nitrogen cycling.
There are two main mechanisms of nonsymbiotic nitrogen fixation:
- Atmospheric Nitrogen Fixation:
- Lightning: Lightning strikes can break the strong nitrogen-nitrogen triple bonds in the atmosphere, creating reactive nitrogen species that eventually combine with water to form nitrates. These nitrates are then deposited onto the soil through rainfall.
- UV Radiation: Ultraviolet (UV) radiation from the sun can also contribute to atmospheric nitrogen fixation. UV radiation dissociates atmospheric nitrogen molecules, leading to the formation of nitrogen oxides, which can dissolve in water and form nitrates.
- Industrial Nitrogen Fixation (Haber-Bosch Process):
- Haber-Bosch Process: Developed in the early 20th century, the Haber-Bosch process is a human-engineered method for fixing nitrogen on an industrial scale. It involves combining atmospheric nitrogen with hydrogen derived from natural gas or other sources at high temperatures and pressure to produce ammonia. This ammonia can then be used as a precursor for nitrogen-based fertilizers.
Nonsymbiotic Nitrogen Fixation Benefits and Drawbacks:
Agricultural Benefits:
- Enhanced Crop Yields: Nitrogen fixation, especially through industrial methods, provides a vital source of nitrogen for agriculture, increasing crop productivity and food production.
- Global Food Security: The ability to produce synthetic nitrogen fertilizers has played a crucial role in sustaining the world’s growing population.
Environmental Concerns:
- Eutrophication: Excess nitrogen from fertilizer runoff can lead to water bodies becoming enriched with nutrients, causing eutrophication, algal blooms, and oxygen depletion.
- Air Pollution: Industrial nitrogen fixation can result in the release of nitrogen oxides, which contribute to air pollution and acid rain.
- Biodiversity Impact: Nitrogen deposition from human activities can disrupt ecosystems, favoring certain plant species over others and altering nutrient dynamics.
Energy and Resource Implications:
- Energy Intensive: Industrial nitrogen fixation is energy-intensive, relying on fossil fuels for hydrogen production. This has environmental and resource implications.
- Carbon Footprint: The energy and emissions associated with nitrogen fertilizer production contribute to carbon emissions and climate change.
Nonsymbiotic nitrogen fixation underscores the complexity of the nitrogen cycle and the interconnectedness of natural and anthropogenic processes. While it offers benefits for agriculture and food security, careful management is crucial to mitigate the environmental impacts and maintain a balance between productivity and sustainability.
Importance of nitrogen for biological processes “short”
The importance of nitrogen in biological processes is profound. Nitrogen is a critical component of proteins, DNA, and energy-carrying molecules like ATP. It’s essential for cell growth, signaling, and photosynthesis in plants.
Nitrogen’s role in nutrient cycling impacts ecosystems and agricultural productivity. From building genetic material to driving energy transfer, nitrogen underpins the fundamental functions of life.
Symbiotic and Nonsymbiotic Nitrogen Fixation comparison chart
Here’s a comparison chart outlining the key differences between symbiotic and nonsymbiotic nitrogen fixation:
| Aspect | Symbiotic Nitrogen Fixation | Nonsymbiotic Nitrogen Fixation |
|---|---|---|
| Definition | Plant-microorganism partnership for nitrogen fixation | Microbial nitrogen fixation independent of plants |
| Partnership Type | The close association between specific plants and microbes | No specific plant-microbe partnership |
| Microorganisms Involved | Rhizobia, Frankia (bacteria) | Free-living bacteria, cyanobacteria, lightning, etc. |
| Nitrogen Source | Atmospheric nitrogen (N2) | Atmospheric nitrogen (N2) |
| Nitrogen Conversion | Converted into ammonia or nitrate by microorganisms | Converted through lightning, UV radiation, or the Haber-Bosch process |
| Examples | Legume-rhizobium association, actinorhizal symbiosis | Lightning, UV radiation, Haber-Bosch process |
| Plant Benefits | Direct access to fixed nitrogen, reduced nitrogen demand | No direct plant-microbe benefits |
| Environmental Impact | Enriches soil fertility, enhances ecosystem health | Can contribute to eutrophication, air pollution |
| Energy Requirements | The plant supplies carbohydrates for microorganisms | Industrial nitrogen fixation requires energy |
| Agricultural Role | Enhances nitrogen availability, sustainable agriculture | Increases crop yields, supports food security |
| Human Influence | Occurs naturally and agriculturally | Involves industrial processes and human intervention |
| Ecosystem Impact | Influences nutrient cycling, soil health, and diversity | Can disrupt ecosystems and nutrient dynamics |
| Resource Implications | Minimal external resource inputs | Requires energy, and fossil fuels for industrial fixation |
| Balance Considerations | Balancing nutrient exchange and competition | Balancing productivity and environmental impacts |
Both forms of nitrogen fixation play essential roles in shaping ecosystems, agricultural productivity, and the global nitrogen cycle.
Factors Influencing Nitrogen Fixation
Factors Influencing Nitrogen Fixation:
- Soil Conditions: Soil pH, moisture levels, and nutrient availability affect the activity of nitrogen-fixing microorganisms.
- Temperature: Optimal temperatures promote microbial activity, but extreme conditions can inhibit nitrogen fixation.
- Cofactors and Minerals: Adequate supplies of minerals like molybdenum and iron are necessary for nitrogen-fixing enzyme function.
- Carbon and Energy Sources: Nitrogen-fixing microorganisms require energy and carbon compounds from host plants or other sources.
- Competition and Inhibition: Competition for resources and inhibitory substances in the soil can affect nitrogen fixation.
- Oxygen Levels: Symbiotic nitrogen fixation requires low oxygen levels to protect nitrogen-fixing enzymes.
- Plant Genetics: Genetic traits of host plants and microbial partners influence the efficiency of nitrogen fixation.
- Symbiotic Specificity: Compatibility between plant hosts and nitrogen-fixing microorganisms varies.
- Environmental Stress: Stressors like pollution or drought can reduce nitrogen fixation rates.
- Human Activities: Agricultural practices, industrial pollution, and land use changes impact nitrogen fixation rates.
Future Directions and Challenges
Future Directions and Challenges:
- Biotechnology: Advancing genetic engineering for more efficient nitrogen-fixing organisms.
- Climate Impact: Understanding how climate change affects nitrogen fixation and ecosystems.
- Sustainable Agriculture: Balancing nitrogen use for food production and environmental protection.
- Nitrogen Management: Developing strategies to reduce pollution from excess nitrogen.
- Microbiome Studies: Exploring microbial interactions for improved nitrogen fixation.
- Technological Innovation: Monitoring and optimizing nitrogen fixation processes.
- Policy and Education: Implementing policies and raising awareness about responsible nitrogen use.
- Global Collaboration: Addressing nitrogen challenges requires international cooperation.
Conclusion
Nitrogen fixation, whether symbiotic or nonsymbiotic, plays a vital role in sustaining life on Earth. It shapes ecosystems, supports agriculture, and underpins essential biological processes. As we navigate challenges and explore future directions, responsible management of nitrogen resources will be crucial for a balanced and sustainable future.