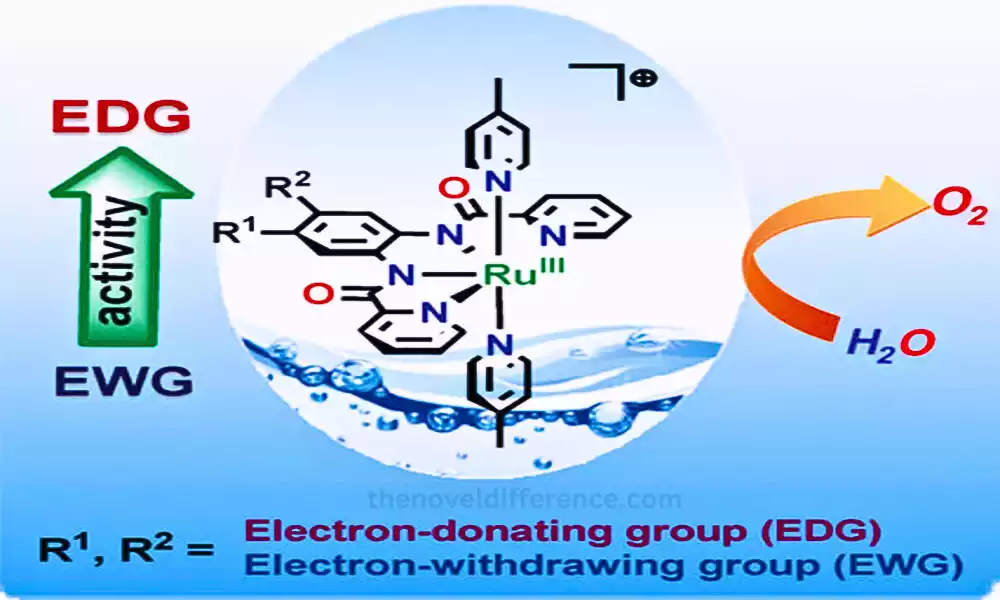
The critical difference between EDG and EWG is that EDG (refers to electron-donating groups). A conjugated pi can increase the electron density of the system where EWG (stands for electron withdrawing groups). A conjugated pi decreases the electron density of the system.
EDG and EWG are electrophilic aromatic directing groups. Both of these are alternative forms that we can find in organic compounds.
Definition of Electron-Donating Groups (EDG)
EDG: Electron-donating groups (EDG) are functional groups or substituents that possess a higher electron density than the surrounding atoms of a molecule. They tend to release or donate electrons toward adjacent atoms or functional groups. Each electron-donating atom is due to the presence of an electron-negative atom (such as oxygen or nitrogen) or pi bonds (double or triple bonds) between groups, which can share electron density.
Definition of Electron-Withdrawing Groups (EWG)
EWG: Electron-withdrawing groups (EWG) are functional groups or substituents that possess higher electronegativity than surrounding atoms in a molecule. These groups tend to withdraw or pull electron density from adjacent atoms or functional groups. The presence of strongly electronegative atoms, such as oxygen or halogen, contributes to the electron-withdrawing nature of these groups.
Importance of EDG
EDG stands for Electron Donating Group. We also know them as “Electron Releasing Group (ERG)”. They act as substitutes for organic compounds that can donate some of their electron density and a conjugated Pi system. This is done through influence or induction effect. This makes the pi-electron system much more nucleophilic.
EDG, when attached to a benzene ring, allows the benzene ring to undergo electrophilic substitution reactions. Because EDG allows the electron density of the benzene ring to increase greatly. Hence this type of electrophilic substitution reaction undergoes.
So EDG can greatly increase the reaction rate. Therefore, we can call these substituents active groups for the aromatic ring. Some examples of EDGs include primary, phenoxides, ethers, secondary, tertiary amines, phenols, etc.
Hydroxyl groups
Hydroxyl groups (-OH) are functional groups that consist of an oxygen atom bonded to a hydrogen atom. They are one of the most important and versatile functional groups in organic chemistry, found in a wide range of compounds. Hydroxyl groups contribute to the unique properties and reactivity of molecules, making them a focal point of study and application.
Key Characteristics and Effects of Hydroxyl Groups:
- Polarity and Hydrogen Bonding: The oxygen atom in the hydroxyl group is more electronegative than hydrogen, creating a polar covalent bond. This polarity makes hydroxyl groups capable of forming hydrogen bonds with other electronegative atoms, such as oxygen or nitrogen. Hydrogen bonding plays a crucial role in determining the physical properties, such as boiling point, solubility, and viscosity, of compounds containing hydroxyl groups.
- Acid-Base Properties: Hydroxyl groups can act as both acids and bases. They are weak acids, as they can donate a proton (H+) from the hydrogen atom. This property contributes to the characteristic acidity of alcohols. Conversely, the oxygen atom can accept a proton, making hydroxyl groups weak bases.
- Stabilization of Positive Charges: Hydroxyl groups stabilize adjacent positive charges by donating electron density through the oxygen atom. This electron donation can influence reaction mechanisms and product formation.
- Nucleophilic Substitution: Hydroxyl groups are nucleophilic and can undergo nucleophilic substitution reactions. In the presence of appropriate reagents, the oxygen atom donates its lone pair of electrons to an electrophilic center, leading to bond formation.
- Reaction with Acids and Dehydration: Hydroxyl groups can react with strong acids to form water and the corresponding salt. Additionally, they can participate in elimination reactions, such as dehydration, where a water molecule is removed to form a double bond.
- Synthesis and Functionalization: Hydroxyl groups are often used as critical intermediates or starting points in organic synthesis. They can be selectively modified through various reactions to introduce other functional groups or achieve specific chemical transformations.
- Biological Importance: Hydroxyl groups are prevalent in biomolecules, such as carbohydrates, alcohol, and some amino acids. They contribute to the structure and function of these molecules, playing essential roles in processes like cell recognition, enzymatic reactions, and DNA binding.
Importance of EWG
EWG is an electron-withdrawing group, which is a functional group that plays an important role in withdrawing electrons from a molecule. It plays an important role in making it less reactive and nucleophilic.
The presence of an EWG in a molecule tends to have various effects on its properties. For example, it destabilizes the conjugate base through delocalization of the negative charge and tends to reduce the acidity of the molecule.
EWG tends to reduce the basicity of the molecule by withdrawing the lone pair of electrons that accept the proton.
Cyano groups
Cyano groups (-CN) are functional groups consisting of a carbon atom triple-bonded to a nitrogen atom. They are electron-withdrawing groups (EWGs) due to the electronegative nature of the nitrogen atom and the presence of the carbon-nitrogen triple bond. Cyano groups are versatile and widely used in various chemical applications, ranging from organic synthesis to medicinal chemistry.
Key Characteristics and Effects of Cyano Groups:
- Electron-Withdrawing Nature: The carbon-nitrogen triple bond in cyano groups firmly withdraws electron density from adjacent atoms or functional groups. This electron-withdrawing effect is a result of the high electronegativity of nitrogen and the presence of pi bonds in the triple bond.
- Stabilization of Negative Charges: Cyano groups stabilize negative charges by withdrawing electrons from the surrounding region. This can reduce the reactivity of negatively charged species, making them more stable.
- Electrophilicity Enhancement: Cyano groups enhance the electrophilicity of molecules by reducing the electron density at nearby sites. This makes the molecule more prone to accept electrons in reactions.
- Acidification of Protons: The presence of the nitrogen atom in cyano groups increases the acidity of protons bonded to adjacent carbon atoms. This can influence reaction pathways and product formation.
- Resonance Effects: The cyano group’s ability to donate electrons through resonance can impact the distribution of charge and stability within a molecule.
- Bioisosteric Replacement: Cyano groups are often used as bioisosteric replacements for other functional groups in drug design. Their electron-withdrawing nature can mimic certain chemical and physical properties, leading to desired biological activity.
- Synthetic Building Blocks: Cyano groups are versatile intermediates in organic synthesis. They can undergo a variety of reactions, such as nucleophilic substitution, reduction, and addition, to yield diverse products.
Difference Between EDG and EWG
| Property | EDG(Electron-Donating Groups) | EWG(Electron-Withdrawing Groups) |
|---|---|---|
| Definition | EDG stands for an electron-donating group | EWG stands for an electron-withdrawing group |
| Effect on Resonance | Increases electron density in pi systems | Decreases electron density in pi systems |
| Effects on Reactivity | Increases the rate of reactions that involve a nucleophilic attack. | Increases the rate of reactions that involve an electrophilic attack. |
| Effects on Molecular Properties | Increases boiling point due to the formation of hydrogen bonds with water molecules. | Decreases boiling point due to the absence of hydrogen bonding |
| Examples | -OH, -NH2, -OR, -NHR | -NO2, -CN, -COOH, -C=O |
Final Word
Both EDG and EWG are groups formed by electrophilic aromatic indicators. They exhibit the opposite function when aromatic rings are added. So, we can say that the key difference between EDG and EWG; EDG is a conjugated pi system, which allows it to increase the electron density. Whereas EWG is a conjugated Pi system, it helps to reduce the electron density.