When it comes to studying the characteristics of mixtures, two terms often arise: azeotropic and zeotropic. These terms refer to different types of mixtures and have distinct properties. We are going to dive into the contrast between azeotropic and zeotropic blends, investigating their definitions, behavior, and applications. By understanding these contrasts, you’ll pick up a more profound knowledge of the world of blends and their interesting properties.
Definition of Azeotropic and Zeotropic Mixture
Azeotropic Mixture: An azeotropic blend, moreover known as an azeotrope, alludes to a blend of two or more fluids that bubbles at a consistent temperature and keeps up a steady composition amid the method of refining. The vapor produced has the same composition as the liquid mixture. Azeotropic mixtures cannot be easily separated into their components by simple distillation because the vapor phase has the same composition as the liquid phase. They exhibit unique properties that differ from those of their pure components.
Zeotropic Mixture: A zeotropic blend, too known as a non-azeotropic blend, alludes to a blend of two or more fluids that bubble over a extend of temperatures and have changing compositions amid refining. Zeotropic mixtures can be separated into their components by fractional distillation. The boiling point of a zeotropic mixture changes as the composition changes, and the vapor produced during distillation has a different composition than the original liquid mixture. Zeotropic mixtures are commonly encountered in various industrial processes and have specific applications in different fields.
Importance of understanding different types of mixtures
Understanding different types of mixtures, such as azeotropic and zeotropic mixtures, is important for several reasons:
1. Separation and purification processes: Different mixtures require different separation techniques. Understanding the characteristics and behavior of azeotropic and zeotropic mixtures allows scientists and engineers to choose the most appropriate separation method. For azeotropic mixtures, alternative separation techniques such as azeotropic distillation or the addition of trainers may be necessary. Zeotropic mixtures, on the other hand, can often be separated through fractional distillation. Knowledge of these distinctions helps in designing efficient separation and purification processes in industries such as chemical manufacturing, petroleum refining, and pharmaceuticals.
2. Industrial applications: Azeotropic and zeotropic mixtures find applications in various industries. For example, azeotropic mixtures are commonly used as solvents, in dehydration processes, and in petroleum refining. Zeotropic mixtures are essential in air separation processes, refrigeration and air conditioning systems, and petrochemical processes. Understanding the behavior and properties of these mixtures enables engineers to optimize their use in industrial applications, leading to improved process efficiency and product quality.
3. Product formulation: Azeotropic and zeotropic mixtures can have unique properties and behaviors that differ from their components. This understanding is crucial for product formulation and development. Industries such as cosmetics, food and beverage, and pharmaceuticals often rely on azeotropic or zeotropic mixtures to create specific product characteristics or achieve desired outcomes. By comprehending the properties and interactions of these mixtures, manufacturers can design products with precise compositions and functionalities.
4. Process optimization and troubleshooting: Knowledge of different mixture types allows for better process optimization and troubleshooting. Understanding the behavior of azeotropic and zeotropic mixtures helps identify potential challenges and develop strategies to overcome them. It enables engineers to adjust operating conditions, modify equipment, or implement alternative separation techniques to improve process efficiency, reduce costs, and ensure product quality.
5. Safety considerations: Different mixtures pose different safety risks. A thorough understanding of azeotropic and zeotropic mixtures helps in assessing and managing these risks. Certain mixtures may be more prone to hazardous reactions or have specific storage and handling requirements. Being aware of their properties allows for appropriate safety measures to be implemented, reducing the potential for accidents, chemical reactions, or environmental hazards.
Understanding diverse sorts of blends, counting azeotropic and zeotropic blends, is fundamental for different logical, building, and mechanical applications. It enables efficient separation processes, optimal product formulation, improved process optimization, and enhanced safety measures.
What are Azeotropic Mixtures?
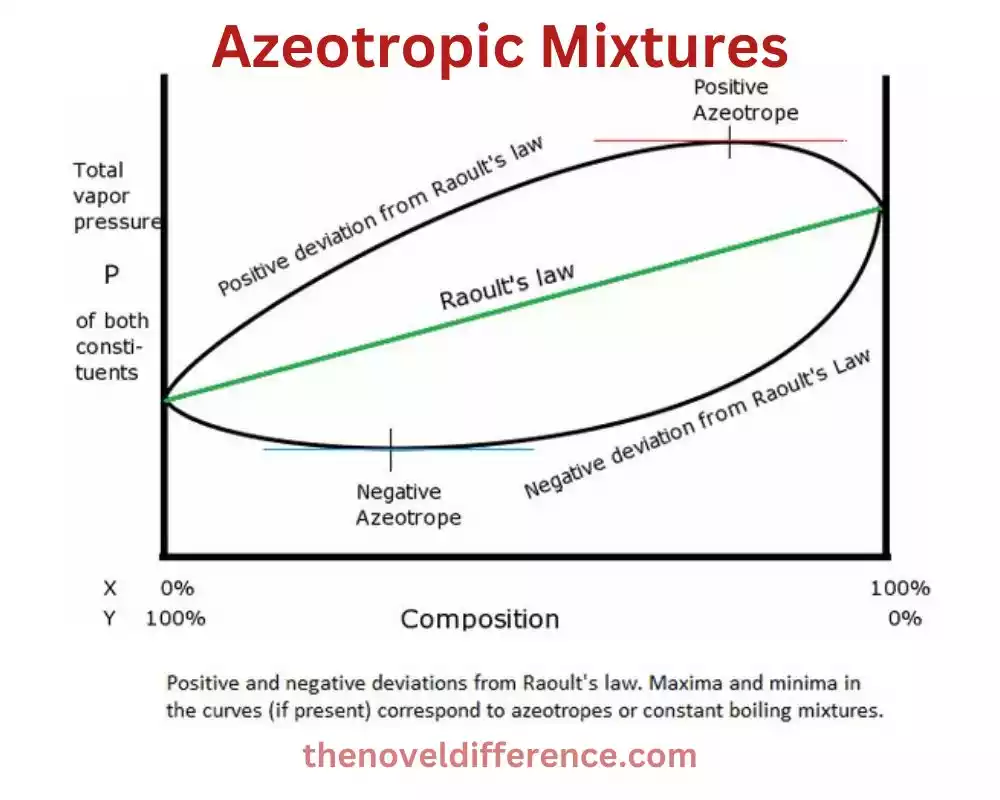
Azeotropic mixtures, also known as azeotropes, refer to mixtures of two or more liquids that exhibit a unique behavior during distillation. In an azeotropic mixture, the liquid components evaporate together at a constant boiling point, and the vapor phase has the same composition as the liquid mixture. This means that during distillation, the composition of the vapor remains constant as it is condensed.
The defining characteristic of an azeotropic mixture is that it cannot be easily separated into its components by simple distillation. This is because the vapor phase maintains the same composition as the liquid phase, making it difficult to achieve a pure separation of the components.
Azeotropic mixtures can be classified into two types:
1. Positive azeotropes: The mixture boils at a lower temperature than any of its components. This means that the vapor phase is richer in the more volatile component compared to the liquid phase. An example of a positive azeotrope is the ethanol-water mixture, which boils at a constant temperature of approximately 78.2 degrees Celsius and has a composition of around 95.6% ethanol.
2. Negative azeotropes: The mixture boils at a higher temperature than any of its components. The vapor phase in a negative azeotrope is typically poorer in the more volatile component compared to the liquid phase. An illustration of a negative azeotrope is the hydrochloric acid-water blend, which shapes an azeotrope with a bubbling point of around 110 degrees Celsius and a composition of around 20% hydrochloric corrosive.
Azeotropic mixtures have various industrial applications. They are often used as solvents, particularly in situations where complete separation of the components is challenging. Azeotropic distillation, where an entrainer is added to modify the composition of the vapor, is employed to separate azeotropic mixtures in certain cases. Additionally, azeotropic mixtures are utilized in processes such as the dehydration of alcohol and the refining of petroleum.
Industrial applications of azeotropic mixtures
Azeotropic mixtures have several important industrial applications due to their unique properties and behavior.
Here are some common industrial applications of azeotropic mixtures:
1. Solvent Extraction: Azeotropic mixtures are often used as solvents in various industrial processes. They can effectively dissolve and extract specific components from mixtures or solid materials. Azeotropic solvents, such as azeotropic alcohol-water mixtures or azeotropic hydrocarbon mixtures, are utilized in processes like the extraction of essential oils, pharmaceutical manufacturing, and chemical synthesis.
2. Dehydration Processes: Azeotropic mixtures are employed in dehydration processes where water needs to be removed from organic compounds or solutions. Azeotropic ethanol-water blends, such as the ethanol-water azeotrope with a composition of around 95.6% ethanol, are commonly utilized within the generation of anhydrous (water-free) ethanol fuel and the pharmaceutical industry for drying out of solvents.
3. Petroleum Refining: Azeotropic mixtures play a crucial role in various stages of petroleum refining processes. They are used in the separation and purification of crude oil fractions, such as the separation of gasoline from other hydrocarbon components. Azeotropic distillation is often employed to break azeotropic mixtures and achieve the desired separation of components.
4. Reactive Distillation: Azeotropic mixtures can be utilized in reactive distillation processes, where chemical reactions occur simultaneously with distillation. The presence of an azeotropic mixture can enhance the reaction kinetics and improve the overall efficiency of the process. Reactive distillation is commonly applied in the production of chemicals, including esters, ethers, and biodiesel.
5. Solvent Recovery: Azeotropic mixtures are also employed in the recovery and recycling of solvents in various industries. When a solvent forms an azeotropic mixture with other substances, such as water or contaminants, azeotropic distillation techniques can be used to separate and recover the pure solvent for reuse. This can be particularly imperative in businesses like pharmaceuticals, paints and coatings, and chemical fabricating, where dissolvable reusing is vital for cost-effectiveness and natural supportability.
6. Chemical Separations: Azeotropic mixtures are sometimes deliberately formed to assist in the separation of closely boiling components in chemical processes. By creating an azeotropic mixture, the boiling points of the components can be shifted, allowing for easier separation through distillation. This technique is utilized in industries such as fragrance production, where the separation of delicate aroma compounds can be challenging.
These are just a few examples of the industrial applications of azeotropic mixtures. The unique properties of azeotropes make them valuable in various separation, purification, and chemical processes across industries.
Solvent extraction processes
Solvent extraction processes, also known as liquid-liquid extraction or simple extraction, involve the separation of components from a mixture using a solvent. This strategy is broadly utilized in different businesses, counting chemical fabricating, pharmaceuticals, nourishment preparation, and mining. Solvent extraction processes utilize the differential solubility of components in immiscible liquid phases to selectively transfer the desired component(s) from one phase to another.
Here is an overview of solvent extraction processes:
1. Principle of Solvent Extraction:
• Solvent extraction relies on the principle that different components in a mixture have different solubilities in different solvents.
• The mixture to be separated, known as the feed or raffinate, is typically a solution or suspension of the desired component(s) in a liquid.
• The solvent used for extraction should be immiscible with the feed and have a preferential affinity for the desired component(s) over the other components.
2. Process Steps:
• Mixing: The feed solution and the solvent are thoroughly mixed to allow mass transfer between the phases. The solute(s) from the feed solution will partition between the two immiscible liquid phases based on their relative solubilities.
• Extraction: During the mixing stage, the desired component(s) preferentially dissolve in the solvent phase, while other components remain in the original phase.
• Phase Separation: The mixture is allowed to settle, and the two liquid phases separate into distinct layers due to their immiscibility. This results in the formation of an organic phase (containing the desired component(s)) and an aqueous or raffinate phase (containing the remaining components).
• Recovery: The organic phase, containing the extracted component(s), is further processed to recover the desired product, often through further separation techniques such as distillation, evaporation, or crystallization. The raffinate phase may undergo additional treatments or be recycled back into the process.
3. Choice of Solvents:
• The selection of solvents depends on the nature of the feed mixture and the desired component(s).
• Common solvents include organic compounds like alcohols, ethers, hydrocarbons, or chlorinated solvents.
• The solvent should have a suitable boiling point, good selectivity for the desired component(s), low toxicity, and economic viability.
4. Industrial Applications:
• Solvent extraction processes find applications in numerous industries, including:
• Chemical Manufacturing: Extraction of specific chemicals, such as separating organic acids from fermentation broths or isolating valuable compounds from reaction mixtures.
• Pharmaceuticals: Isolation and purification of active pharmaceutical ingredients (APIs) from complex mixtures or natural sources.
• Mining and Metallurgy: Extraction of metals from ores or leach solutions.
• Food Processing: Removal of undesirable substances, such as the decaffeination of coffee or the removal of fats and oils from food products.
• Environmental Remediation: Removal of pollutants or contaminants from soil, water, or air.
Solvent extraction processes offer a versatile and effective means of separating components from complex mixtures. They provide high selectivity, allow for large-scale operations, and can be tailored to specific separation requirements across various industries.
Petroleum refining
Petroleum refining, too known as oil refining, could be a complex mechanical preparationAmid fragmentary refining, the blend is warmed, and the vapor that’s created is condensed and collected. That includes the transformation of unrefined oil into different important items such as gasoline, diesel fuel, fly fuel, oils, and petrochemical feedstocks. The refining process utilizes different techniques and unit operations to separate, convert, and purify the components present in crude oil.
Here is an overview of the petroleum refining process:
1. Distillation:
• Crude oil is first subjected to fractional distillation in a distillation column.
• The crude oil is heated, and the vapors rise the column, condensing at different heights based on their boiling points.
• Different fractions, such as gasoline, diesel, kerosene, and heavy residues, are obtained at different levels of the distillation column.
2. Conversion Processes:
• Conversion processes are employed to convert heavier, less desirable fractions into more valuable products.
• Catalytic cracking breaks down heavy hydrocarbons into lighter, more useful fractions like gasoline and diesel.
• Hydrocracking and hydrotreating involve the use of hydrogen under high pressure and temperature to break down heavy hydrocarbons and remove impurities like sulfur, nitrogen, and metals.
3. Reforming:
• Reforming is a process that converts low-octane naphtha into higher-octane gasoline components.
• It involves the use of catalysts and high temperatures to rearrange the hydrocarbon molecules, resulting in improved fuel properties.
4. Desulfurization:
• Desulfurization processes, such as hydrodesulfurization (HDS), are employed to remove sulfur compounds from petroleum products.
• Sulfur compounds in fuels contribute to air pollution and harm the environment. Desulfurization reduces sulfur content to meet regulatory requirements and improve fuel quality.
5. Blending:
• Blending involves mixing different refined products to meet specific quality and performance requirements.
• Gasoline, diesel, and other products may be blended with additives to enhance properties like octane rating, lubricity, stability, and corrosion resistance.
6. Additional Processes:
• Other refining processes include isomerization, alkylation, polymerization, and coking, which are utilized to produce specific products or modify certain properties of the fuels and feedstocks.
7. Environmental Considerations:
• Petroleum refining involves environmental considerations to minimize emissions and comply with regulations.
• Technologies such as catalytic converters, sulfur recovery units, and advanced emissions control systems are employed to reduce pollutants and greenhouse gas emissions.
The petroleum refining industry plays a critical role in meeting global energy demands and providing various refined products. It requires advanced technologies, efficient operations, and continuous research and development to optimize production, meet quality specifications, and ensure environmental sustainability.
What are Zeotropic Mixtures?
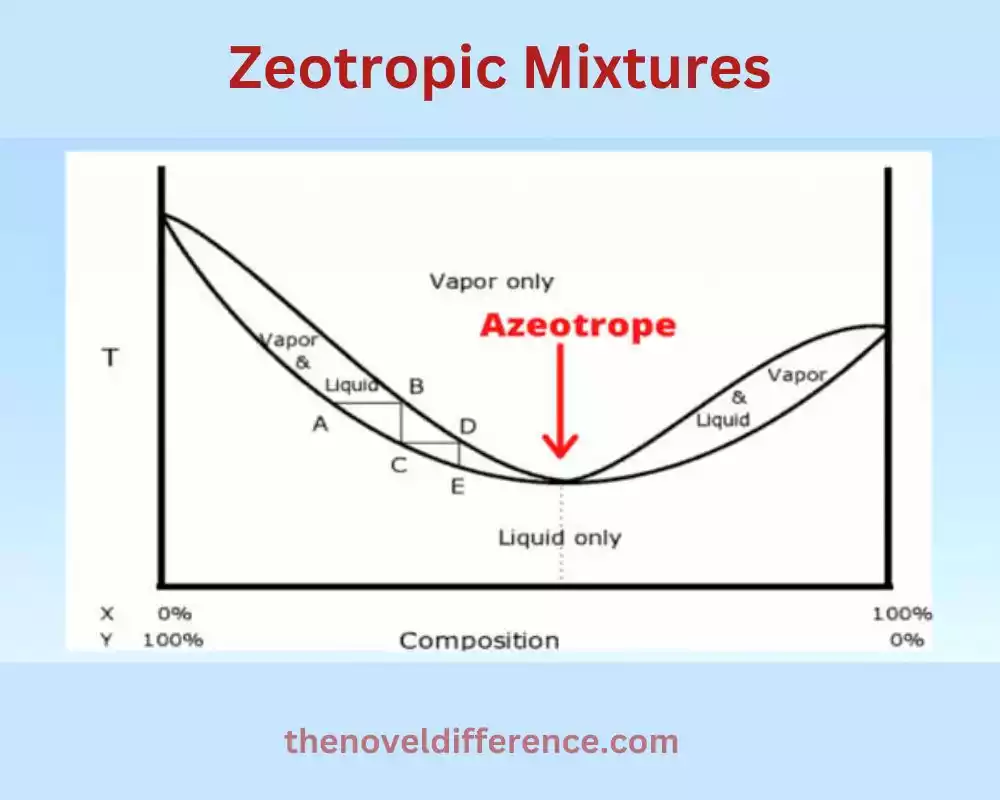
Zeotropic mixtures, also known as non-azeotropic mixtures, are mixtures of two or more substances that exhibit varying compositions and boiling points during distillation. Unlike azeotropic mixtures, zeotropic mixtures can be separated into their components through fractional distillation because the composition of the vapor phase differs from the liquid phase.
Here are some key characteristics of zeotropic mixtures:
1. Variable Boiling Points: Zeotropic mixtures do not boil at a constant temperature like azeotropic mixtures. Instead, they have boiling points that change as the distillation process progresses. The boiling points of the components within the mixture depend on their vapor pressures and interactions with each other.
2. Composition Changes: As zeotropic mixtures are distilled, the composition of the vapor phase differs from the composition of the liquid mixture. This difference in composition allows for the separation of the components through fractional distillation. The composition of the vapor phase typically becomes richer in the more volatile component(s) compared to the liquid phase.
3. Separation through Fractional Distillation: Zeotropic mixtures can be separated into their components by using fractional distillation techniques. During fractional distillation, the mixture is heated, and the vapor that is produced is condensed and collected. The condensed liquid contains a different composition than the original mixture. By collecting different fractions at different temperature ranges, the separation of the components is achieved.
4. Application in Industrial Processes: Zeotropic mixtures find various applications in industrial processes. For example, they are commonly encountered in air separation processes, such as the production of nitrogen, oxygen, and argon through cryogenic distillation. Zeotropic mixtures are also relevant in refrigeration and air conditioning systems, where different refrigerants with specific boiling points and compositions are used.
Understanding the behavior and properties of zeotropic mixtures is crucial for designing separation processes, optimizing industrial operations, and ensuring product quality. Fractional distillation is the primary method used to separate zeotropic mixtures, allowing for the extraction of specific components based on their varying boiling points and compositions.
Characteristics of zeotropic mixtures
Zeotropic blends, moreover known as non-azeotropic blends, show a few key characteristics that recognize them from azeotropic blends.
Here are the main characteristics of zeotropic mixtures:
1. Variable Composition: Zeotropic mixtures have varying compositions throughout the distillation process. The composition of the liquid phase changes as different components evaporate at different rates. The composition of the vapor stage contrasts with that of the fluid stage. This variable composition allows for the separation of components during distillation.
2. Variable Boiling Points: Unlike azeotropic mixtures that have a constant boiling point, zeotropic mixtures display changing boiling points throughout distillation. Each component within the mixture has its vapor pressure, which determines its boiling point. As the distillation progresses, the boiling point of the mixture changes due to the shifting composition.
3. Separation Potential: Zeotropic mixtures can be separated into their components using fractional distillation. By exploiting the differences in boiling points and compositions, it is possible to collect different fractions at various temperature ranges, thereby isolating the desired components.
4. Composition Enrichment: During fractional distillation of zeotropic mixtures, the composition of the vapor phase tends to be enriched in the more volatile component(s) compared to the liquid phase. This enrichment is a result of the preferential evaporation of the more volatile component(s) during the distillation process.
5. Phase Diagram Complexity: The phase diagrams of zeotropic mixtures are more complex than those of azeotropic mixtures. Zeotropic mixtures typically exhibit a boiling point curve on the phase diagram that represents the temperature-composition relationship during distillation. This curve shows how the boiling point changes as the composition of the liquid and vapor phases evolves.
6. Application-Specific Behavior: The behavior of zeotropic mixtures can vary depending on the specific components and their interactions. The separation efficiency and ease of separation may differ for different zeotropic mixtures. Understanding the thermodynamic properties and interactions of the components is crucial for optimizing separation processes and designing efficient distillation systems.
These characteristics of zeotropic mixtures highlight their dynamic nature during distillation and the potential for separating components based on their varying boiling points and compositions. Fractional distillation is the primary technique used to exploit these characteristics and separate zeotropic mixtures into their components.
Boiling point range
The bubbling point run alludes to the temperature run over which the blend bubbles and condenses amid the refining handle. The boiling point range is a result of the varying compositions of the mixture as different components evaporate and condense at different temperatures.
The boiling point range of a zeotropic mixture is determined by the boiling points of its components and their relative concentrations in the mixture. As the distillation process progresses, the boiling point of the mixture changes due to the shifting composition. The boiling point range can be visualized on a temperature-composition diagram or a distillation curve.
The width of the boiling point range can vary depending on the specific zeotropic mixture. Some zeotropic mixtures may have a relatively narrow boiling point range, while others may have a broader range. The bubbling point run can be affected by variables such as the instability of the components, their intuition, and the general composition of the blend.
During distillation, different fractions of the zeotropic mixture are collected at different temperature ranges within the boiling point range. Each fraction represents a different composition and may contain specific components of interest. Fractional distillation allows for the separation of components by selectively collecting fractions based on their boiling points.
It is important to note that the boiling point range of a zeotropic mixture is different from the constant boiling point exhibited by azeotropic mixtures. Azeotropic mixtures have a single, constant boiling point, while zeotropic mixtures exhibit a range of boiling points due to their changing composition.
Variable composition during distillation
Variable composition during distillation is a characteristic of zeotropic mixtures. Zeotropic mixtures consist of two or more components that have different boiling points and exhibit varying compositions as they undergo the distillation process.
During distillation, the zeotropic mixture is heated, causing the more volatile component(s) to evaporate first. As the vapor rises in the distillation column, it condenses at different heights based on its composition and condenses into a liquid phase. The liquid phase, known as the distillate, has a different composition from the original mixture.
The changing composition occurs because different components in the mixture have different vapor pressures and boiling points. Components with lower bubbling focuses dissipate more promptly and tend to concentrate within the vapor stage, whereas components with higher boiling focuses stay within the fluid stage. This leads to a progressive change in the composition of the liquid and vapor phases as distillation proceeds.
As a result of the variable composition, divisions of diverse compositions can be collected at distinctive stages of the refining handle. This allows for the separation and isolation of individual components or groups of components from the zeotropic mixture.
The ability to manipulate the composition of the zeotropic mixture during distillation is essential for various industrial applications. It enables the production of specific products with desired purity, the removal of impurities, and the separation of valuable components from complex mixtures.
Understanding the variable composition behavior of zeotropic mixtures is crucial for designing effective distillation processes, optimizing separation efficiency, and achieving desired product specifications.
Difference between Azeotropic and Zeotropic Mixtures
The main difference between azeotropic and zeotropic mixtures lies in their behavior during distillation and their ability to be separated into their components.
Here are the key differences between azeotropic and zeotropic mixtures:
1. Boiling Point Behavior:
• Azeotropic Mixtures: Azeotropic mixtures have a constant boiling point at a specific composition. When distilled, the vapor phase has the same composition as the liquid phase, making it difficult to separate the components by simple fractional distillation.
• Zeotropic Mixtures: Zeotropic mixtures have varying compositions and boiling points during distillation. The composition of the vapor phase differs from the liquid phase, allowing for the separation of the components through fractional distillation.
2. Separation Potential:
• Azeotropic Mixtures: Azeotropic mixtures cannot be easily separated into their components by simple fractional distillation since the composition of the vapor phase remains constant. Additional separation techniques, such as azeotropic distillation or the use of trainers, are required to separate the components.
• Zeotropic Mixtures: Zeotropic mixtures can be separated into their components using fractional distillation. The varying compositions during distillation enable the selective collection of different fractions at different temperature ranges, facilitating the separation of components.
3. Constant vs. Variable Composition:
• Azeotropic Mixtures: Azeotropic mixtures have a constant composition in both the liquid and vapor phases at the azeotropic point. The composition does not change during distillation.
• Zeotropic Mixtures: Zeotropic mixtures have a variable composition in both the liquid and vapor phases during distillation. The composition changes as different components evaporate at different rates, allowing for separation.
4. Industrial Applications:
• Azeotropic Mixtures: Azeotropic mixtures are commonly encountered in industrial applications such as solvent recovery, where the separation of an azeotropic mixture requires additional techniques such as azeotropic distillation or molecular sieves.
• Zeotropic Mixtures: Zeotropic mixtures are utilized in various industrial processes, including petroleum refining, air separation, and chemical production. Fractional distillation is employed to separate the components of zeotropic mixtures based on their varying boiling points and compositions.
Understanding the distinction between azeotropic and zeotropic mixtures is crucial in designing separation processes and selecting appropriate techniques to achieve the desired separation goals. While azeotropic mixtures pose challenges for simple fractional distillation, zeotropic mixtures offer the potential for component separation through fractional distillation due to their variable compositions and boiling points.
Comparison Chart
Sure! Here’s a comparison chart highlighting the main differences between azeotropic and zeotropic mixtures:
| Azeotropic Mixtures | Zeotropic Mixtures | |
|---|---|---|
| Boiling Point Behavior | Constant boiling point at a specific composition | Varying boiling points during distillation |
| Separation Potential | Difficult to separate components by simple fractional distillation | Separable through fractional distillation |
| Constant vs. Variable Composition | Constant composition in both liquid and vapor phases | Variable composition in both liquid and vapor phases |
| Separation Techniques | Require additional techniques like azeotropic distillation or entertainers for separation | Separable by fractional distillation |
| Industrial Applications | Common in solvent recovery processes | Used in various industries such as petroleum refining, air separation, and chemical production |
Please note that this chart provides a general overview of the differences between azeotropic and zeotropic mixtures, but there may be specific cases or exceptions depending on the individual mixture and its components.
Conclusion
The difference between azeotropic and zeotropic mixtures lies in their behavior during distillation. Azeotropic mixtures maintain a constant boiling point and composition, while zeotropic mixtures exhibit varying boiling points and compositions.
Each type of mixture has its own set of applications and challenges. By understanding these differences, scientists and engineers can utilize these unique properties to their advantage in various industries. Whether it’s the creation of alcoholic beverages or the production of refrigerants, the world of azeotropic and zeotropic mixtures offers a fascinating realm of possibilities.
