Overview of CHO cells
Chinese Hamster Ovary cells (CHO) are derived from the Cricetulus grseus, a Chinese hamster.
CHO cells are one of the most commonly used mammalian cellular lines in biotechnology. Here’s an overview of the CHO cell:
- Origin and History: In the 1950s, CHO cells were isolated from the ovary tissues of Chinese Hamsters. Since then, these cells have been studied extensively, characterized, and developed into robust lines of cells for various applications.
- Cell Line Characteristics: CHO cells are adherent cells, meaning they require a solid surface for growth. However, some CHO cell lines, such as CHO-S, have been adapted to suspension culture, allowing them to grow in suspension without the need for a solid surface.
- Growth Properties: CHO cells are robust and have a rapid growth rate. They can proliferate rapidly under appropriate culture conditions and achieve high cell densities. CHO cells can be cultured in a variety of media formulations optimized for their growth and productivity.
- Protein Expression: CHO cells are known for their ability to express recombinant proteins at high levels. They possess a well-developed protein synthesis and secretion machinery, making them capable of producing complex proteins with proper folding and post-translational modifications.
- Post-Translational Processing: CHO cells are able to perform post-translational processing, such as glycosylation and phosphorylation. CHO cells can add complex sugar chains to proteins. This can have a significant impact on functionality, stability, and immunogenicity.
- Genetic Stability: CHO cells exhibit genetic stability and maintain the expression of introduced genes over multiple generations. This stability is crucial for the consistent and reliable production of recombinant proteins.
- Bioproduction Applications: CHO cells are used to produce a wide range of biopharmaceuticals including monoclonal antibodies, therapeutic proteins and enzymes. Due to their high expression of proteins, scalability, and regulatory acceptance, they are ideal for large-scale production.
- Cell Line Engineering: CHO cells can be genetically engineered and modified to enhance protein expression, improve product quality, or introduce specific traits. This genetic engineering potential allows researchers to develop customized CHO cell lines tailored to specific bioproduction requirements.
CHO cells have revolutionized the biopharmaceutical industry by serving as a versatile and reliable platform for the production of complex therapeutic proteins. Their robust growth, high protein expression levels, post-translational modification capabilities, and genetic stability have made them the preferred choice for large-scale bioproduction processes.
Introduction to the CHO-S and CHO-K1 cell line
Chinese Hamster Ovary cells (CHO) are widely used in biotechnology and biomedical studies to produce therapeutic proteins, vaccines and other biopharmaceuticals. CHO cells have many advantages, including high protein expression and post-translational modification capabilities. They are also compatible with large-scale manufacturing processes.
The two most commonly used CHO cells lines are CHO K1 and CHO S. They have different characteristics that make them appropriate for various applications and research purposes. Understanding the differences between CHO S and CHO K1 is important for selecting the correct cell line based on specific experimental requirements.
This content outline will explore the characteristics, genetic background, growth properties and applications of CHO-S versus CHOK1 cell lines. We can learn more about the capabilities and unique characteristics of each cell by exploring these factors. We will be able to make better decisions about their use for biotechnological purposes.
Importance of CHO cells in Biotechnology
Due to their exceptional characteristics and versatility, CHO cells are of great importance in biotechnology.
Here are some of the main reasons why CHO cell applications are so valuable:
- Protein Expression: CHO Cells have the ability of producing complex and high quality recombinant protein. They have the cellular machinery necessary for folding, post-translational modification (such as glycosylation), or secretion of protein. CHO cells are therefore ideal for producing therapeutic proteins, enzymes and hormones.
- Scalability: CHO cells can be easily adapted to large-scale production processes. They can be cultured in bioreactors and scaled up to produce substantial quantities of desired proteins, meeting the demands of the biopharmaceutical industry.
- Safety: CHO cells have a long history of safe usage in bioproduction. They are non-pathogenic and do not harbor human viruses, reducing the risk of contamination in therapeutic protein manufacturing. This makes them highly suitable for the production of biologics intended for human use.
- Regulatory Approval: CHO cells have received regulatory approval by regulatory agencies such as the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA) for the production of various biopharmaceuticals. This regulatory acceptance streamlines the approval process for products derived from CHO cells.
- Genetic Stability: CHO cells exhibit genetic stability, making them suitable for long-term cultivation and consistent production of recombinant proteins. They can maintain stable expression profiles over multiple generations, ensuring reliable and consistent protein production.
- Post-Translational Modifications: CHO cells possess machinery that allows for the appropriate and diverse post-translational modifications of proteins, including glycosylation. These modifications can significantly impact the efficacy, stability, and immunogenicity of therapeutic proteins, making CHO cells an ideal choice for producing biopharmaceuticals with desired glycosylation patterns.
- Cell Line Engineering: CHO cells are amenable to genetic manipulation and engineering. They can be modified to enhance protein expression levels, improve product quality, or introduce specific traits. This genetic engineering potential enables researchers to tailor CHO cell lines for specific bioproduction needs.
The combination of robust protein expression capabilities, scalability, safety, regulatory acceptance, genetic stability, and post-translational modification abilities makes CHO cells indispensable in biotechnology.
Their extensive utilization has revolutionized the production of therapeutic proteins and biopharmaceuticals, facilitating advancements in medicine and healthcare.
CHO-S Cell Line
The CHO-S cell line is a specific variant of Chinese Hamster Ovary (CHO) cells that has been adapted for suspension culture, allowing it to grow and proliferate in a suspension state without the need for a solid surface.
Here is an overview of the CHO-S cell line:
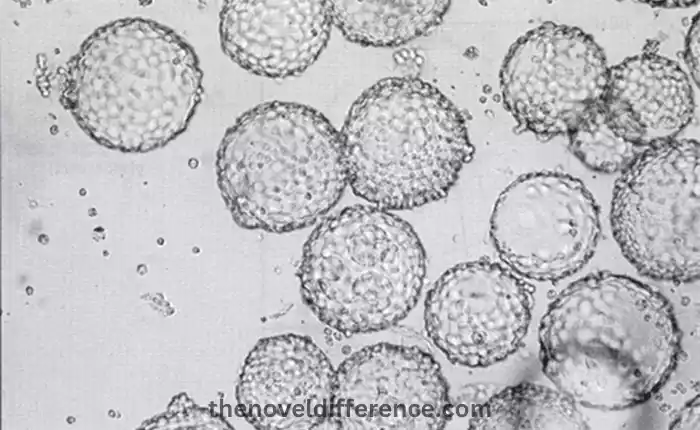
- Characteristics and Origin:
-
- CHO-S cells are derived from the ovaries of Chinese hamsters (Cricetulus griseus).
- They are a subline of CHO cells that have been specifically adapted for suspension culture.
- CHO-S cells are widely used in biopharmaceutical research and production due to their ability to grow in large-scale suspension cultures.
- Growth Properties and Requirements:
-
- CHO-S cells exhibit robust growth and can achieve high cell densities in suspension culture.
- They require a suitable growth medium that provides essential nutrients, vitamins, amino acids, and growth factors for their proliferation.
- CHO-S cells are typically grown in bioreactors or shake flasks, where they can be agitated to maintain suspension and optimal oxygen and nutrient availability.
- Genetic Background and Modifications:
-
- CHO-S cells have a similar genetic background to other CHO cell lines.
- They can be genetically modified and engineered to enhance protein expression, improve product quality, or introduce specific traits.
- The genetic stability of CHO-S cells allows for consistent and reliable expression of recombinant proteins.
- Applications and Uses:
-
- Production of Therapeutic Proteins: CHO-S cells are extensively used for the production of recombinant proteins, including therapeutic antibodies, enzymes, hormones, and other biopharmaceuticals. Their suspension culture adaptability makes them suitable for large-scale protein production.
- Vaccine Development: CHO-S cells are employed in the production of viral vaccines, such as recombinant viral vectors or subunit vaccines, where suspension culture is advantageous for scalable vaccine production.
- Gene Expression Studies: CHO-S cells serve as a valuable model system for studying gene expression, protein synthesis, and post-translational modifications. They are utilized to investigate the effects of various factors on protein production and optimize expression systems.
- Bioproduction of Recombinant Proteins: CHO-S cells are utilized in the bioproduction industry for the large-scale manufacturing of therapeutic proteins, meeting the demand for biologics in healthcare.
The CHO-S cell line’s ability to grow in suspension culture and its robust protein expression capabilities make it a popular choice for biopharmaceutical research and production. Its adaptation to suspension culture allows for scalable production, making it suitable for large-scale bioproduction processes.
CHO-K1 Cell Line
The CHO K1 cell line is a variant of Chinese Hamster Ovary cells (CHO), which has been extensively used in biotechnology, biomedical research and other fields.
This is a brief overview of the CHO K1 cell line:
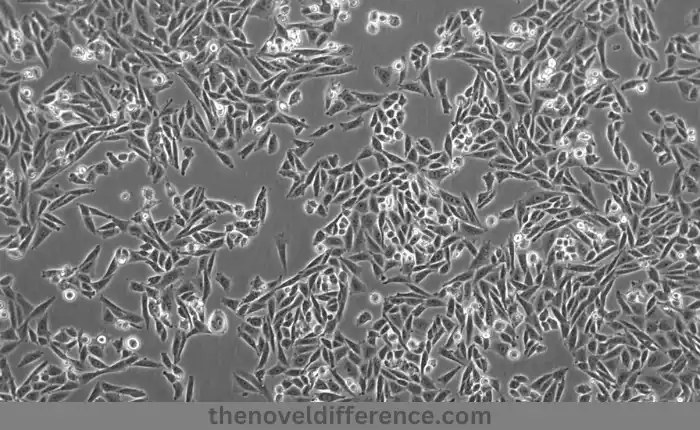
- Characteristics and Origin:
-
- The ovaries of Chinese Hamsters (Cricetulus graus) are used to produce CHO-K1 cell lines.
- They are one of the earliest and most extensively studied CHO cell lines, with a long history of usage in biotechnological applications.
- CHO-K1 cells are adherent cells, meaning they require a solid surface for growth and proliferation.
- Growth Properties and Requirements:
-
- CHO-K1 cells exhibit robust growth and can reach high cell densities when cultured on a suitable solid surface.
- They require a growth medium that provides essential nutrients, vitamins, amino acids, and growth factors for their proliferation.
- CHO-K1 cells are typically cultured in cell culture flasks or dishes, where they adhere to the surface and form a monolayer.
- Genetic Background and Modifications:
-
- CHO-K1 cells have a well-established genetic background and are commonly used as the parental cell line for generating genetically modified CHO cell lines.
- They can be genetically engineered to enhance protein expression, improve product quality, or introduce specific traits using techniques such as transfection and gene knockout.
- Applications and Uses:
-
- Protein Expression and Production: CHO-K1 cells are widely employed for the production of recombinant proteins, including therapeutic proteins, antibodies, enzymes, and other biopharmaceuticals. Their robust protein expression capabilities make them suitable for protein production applications.
- Transient Gene Expression: CHO-K1 cells are often utilized for transient gene expression studies, where genes of interest are transiently transfected into the cells to achieve short-term protein expression for research purposes.
- Gene Editing and Knockout Studies: The well-characterized genetic background of CHO-K1 cells makes them suitable for gene editing techniques, such as CRISPR-Cas9, allowing for the targeted modification or knockout of specific genes to study their function or impact on protein production.
- Cell Line Development: CHO-K1 cells serve as a starting point for the development of stable and high-producing cell lines. They can be used as the parental cell line for generating clones with desired characteristics and enhanced protein expression.
The CHO-K1 cell line’s robust growth, well-established genetic background, and versatility in protein expression make it a valuable tool in biotechnological research. Its adherent nature makes it suitable for applications that require a solid surface for cell growth and protein production.
CHO-S and CHO-K1 in Tabular Form
CHO-S and CHO-K1 are cell lines derived from the ovary of the Chinese hamster. They are widely used in biological and pharmaceutical research.
You can find a comparison of these two cell lines in tabular form:
| Feature | CHO-S | CHO-K1 |
|---|---|---|
| Origin | Chinese Hamster Ovary | Chinese Hamster Ovary |
| Primary Use | Protein Production | Protein Production |
| Growth Characteristics | Suspension Culture | Adherent Culture |
| Genetic Characteristics | DHFR- (Dihydrofolate Reductase) | DHFR+ (Dihydrofolate Reductase) |
| Mutations | Specific mutations to DHFR | Generally wild-type |
| Typical Applications | Therapeutic Protein Expression | Research & Development, Therapeutic Protein Expression |
CHO-S cells are specifically engineered to be dihydrofolate reductase (DHFR) deficient, which makes them suitable for a particular method of gene amplification. CHO-K1 cells are more similar to the wild-type and are commonly used in general research and development.
What are the Similarities Between CHO-S and CHO-K1
CHO-S cells and CHOK1 cells are both distinct variations of Chinese Hamster Ovary Cells (CHO). They each have their own unique features, but they also share some similarities.
There are several similarities between the CHO-S cell line and the CHOK1 cell line:
- Chinese Hamster Ovary Origin: Both the CHO-S cell line and the CHOK1 cells are derived directly from ovaries taken from Chinese hamsters. Both cell lines are from the same species, and they have similar genetic backgrounds.
- Mammalian Cell Lines: CHO-S and CHO-K1 cells are mammalian cell lines commonly used in biotechnology and biomedical research. As mammalian cells, they share similar cellular machinery, including protein synthesis and secretion pathways.
- Protein Expression Capabilities: Both CHO-S and CHO-K1 cells are known for their robust protein expression capabilities. They possess the cellular machinery necessary for the production of recombinant proteins and can efficiently fold and process these proteins.
- Genetic Manipulation: CHO-S and CHO-K1 cells can be genetically modified and engineered to enhance protein expression, improve product quality, or introduce specific traits. Techniques such as transfection and gene knockout can be applied to both cell lines for genetic manipulation.
- Biopharmaceutical production: Both CHO S and CHO K1 cell lines are widely used to produce biopharmaceuticals such as therapeutic proteins, enzymes and vaccines. These cells are used in large-scale production processes to meet biopharmaceutical demands.
- Research Tools: CHO-S and CHO-K1 cells serve as valuable tools for research purposes. They are utilized for studying gene expression, protein synthesis, post-translational modifications, and other cellular processes. Researchers often compare and contrast the characteristics of these two cell lines to gain insights into protein production and cell behavior.
While there are differences between CHO-S and CHO-K1 cells, these similarities highlight their shared applications and relevance in biotechnology and biomedical research. Both cell lines have contributed significantly to advancements in protein production, drug development, and understanding of cellular processes in mammalian systems.
Conclusion
CHO-S and CHO-K1 cell lines represent two variants of Chinese Hamster Ovary (CHO) cells with distinct characteristics that make them ideal for various biotechnology and biomedical research applications.
CHO-S cells have been tailored for suspension culture, enabling them to grow and proliferate without the need for solid surfaces. These high-density growth suspension cells are commonly used for large-scale production of recombinant proteins as they have excellent protein expression capabilities with specific glycosylation patterns.
CHO-S cells also excel as an economical bioproduction option with specific glycosylation patterns allowing scalable production processes with ease.
CHO-K1 cells are adherent cells that require a solid surface for growth and form a monolayer when cultured. Their genetic background has long been established, making CHO-K1 an excellent parent cell line from which genetically modified lines may be generated.
Furthermore, their versatility extends to protein expression studies, transient gene expression studies, gene editing applications, and stable cell line development projects.
